Long-haul transportation in trucks and ships, steel manufacturing, chemical synthesis… What do all these economic sectors have in common? Well, at least two things: first, it’s very challenging to reduce their CO2 emissions; and, most importantly, green hydrogen could play an important role in their decarbonisation. However, there’s hurdles ahead: we need to produce hydrogen at an industrial scale, and this presents several challenges. How do we actually make hydrogen? Where do we get this key element? This explainer focuses on electrolysis, a key method to get green hydrogen, and the technology that makes it possible – electrolysers.
Electrolysis: getting green hydrogen from water
Water covers over two thirds of the Earth’s surface. And, since centuries ago, we know it’s made of two elements: hydrogen and oxygen. Therefore, it seems like the perfect candidate to provide us with the hydrogen we need. We just have to split the water molecule into its fundamental components. But, how could we do that? It’s also well known – we use electricity, and a process called electrolysis.

As many innovations in chemistry, electrolysis was discovered by pure chance. It was first observed by two Dutch merchants, Jan Rudolph Deiman and Adriaan Paets van Troostwijk, back in 1789. Then, in the early nineteenth century, William Nicholson and Anthony Carlisle were closely looking into Volta’s experiments with electricity. They attached two wires to both poles of a battery, submerged them in a tube filled with water… and noticed something. Around the submerged wires, bubbles started to pop – one stream at double the rate than the other. You’ve guessed it right, the bubbles were hydrogen and oxygen. Soon, electrolysis emerged as a fashionable field in science, mostly used to isolate new chemical elements.
Since then, the technology for water splitting has advanced a lot, growing beyond just running a continuous current of electricity through water. Nowadays, electrolysis takes place in industrial machines called electrolysers. Let’s investigate the different types of electrolysers and how they work.
The who is who of electrolysers
To make electrolysis happen, we just need three ingredients:
- Water: our main protagonist. Sadly, not every type of water is “electrolyser-ready”. Electrolysers today need something called ultrapure water, which has been purified – cleaned of contamination and dirt, to ensure the electrolyser runs smoothly. Ultrapure water also reduces the degradation and wear of the materials over time. Oddly enough, there’s not a scientific and specific definition of ultrapure water, as the parameters vary depending on the different electrolysers. At ANEMEL, we’re trying to avoid this step altogether, producing a technology for electrolysers that work directly with dirty waters, such as wastewater and seawater.
- Electricity. As we explained in our previous post, hydrogen is codenamed with different colours (green, blue, grey…), depending on the environmental impact and carbon emissions of its production method. When we use electricity from renewable sources to feed the electrolyser, we make green hydrogen – with virtually zero carbon emissions.
- An electrolyte. This is a material that contains ions (that’s just electrically charged particles), and therefore it’s able to conduct electricity by moving the ions around. This, maybe surprisingly, conducts electrical current without moving electrons around.

The design of electrolysers allows for a high modularity and scalability. The basic unit of an electrolyser is called a “cell”, which consists of an anode, a cathode and a membrane. In an electrolytic cell, electricity goes in through the cathode, and comes back to the circuit via the anode. Then, cells are assembled together, in what’s known as a “cell stack” or, simply, a “stack”, which produces more hydrogen (and oxygen) as the number of cells increases. When we apply an electrical current to the stack, we split water to form hydroxide ions (OH–), which move through the electrolyte towards the anode. The remaining hydrogen atom combines with electrons from the electrical current to form hydrogen gas – while oxygen gas bubbles on the anode.
However, the devil is in the details, and there’s multiple ways of mixing these three basic ingredients. When we scale up to industrial applications, we find many types of electrolysers. Normally, they’re classified based on the materials used as an electrolyte – the part that moves the charged particles around. Depending on that, there’s four different types of electrolysers: alkaline, solid oxide, anion exchange membrane and proton exchange membranes. Some of these technologies are already commercially available, while some others are still maturing (head directly to the final section if you want to know more on that), but there’s plenty of room for improvement in all of them, thank to projects like ANEMEL.
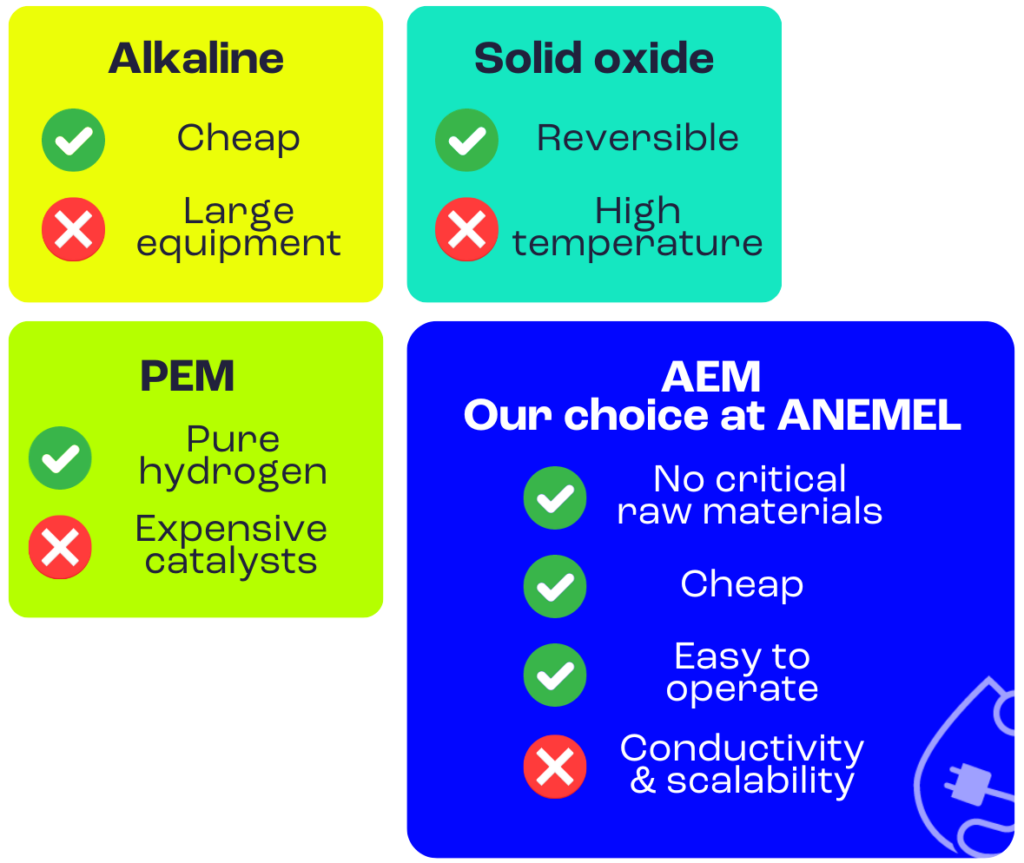
Alkaline electrolysers
This type of electrolyser work at low temperature, using alkaline solutions (with substances like sodium hydroxide and potassium hydroxide, for example). It’s the most mature of the technologies available, and also currently the cheapest. However, it has some downsides: the equipment needed is quite large, and the hydrogen produced needs some further treatments and purifications before it’s used, which increases the cost.
Solid oxide electrolysers (SOEC)
These electrolysers have a different approach – they use an electrolyte made from low-cost ceramic materials. Their main advantage is reversibility: they produce hydrogen from electricity, but also could consume hydrogen to produce electricity, like a fuel-cell. Although they reach a high degree of energy efficiency, they also face huge challenges. For example, they need extremely high temperatures to operate, between 700 and 850 ºC. This is not only hard to achieve and maintain, but also affect the materials of the electrolyser, compromising durability and stability, and lead to unwanted carbon emissions derived from the heating process.
Anion exchange membrane (AEM) electrolysers
It uses a membrane as an electrolyte, to conduct anions. AEM have a few advantages: they don’t use as many critical materials as the other technologies, meaning they’re usually cheaper and easier to operate. However, they still have to improve their efficiency, conductivity and stability.
AEMs is the type of technology we’re developing at ANEMEL. One of our main goals is, precisely, putting the problem of expensive metals away. To make water splitting more sustainable, catalysts could come from abundant elements, such as iron and nickel. That’s precisely one of the goals of our project: we want to develop AEM electrolysers that work well with abundant metals, to make hydrogen production greener, and also cheaper.
Proton exchange membrane (PEM) electrolysers
PEMs also use a membrane to exchange protons electrolyte, but they need slightly acidic conditions to work at low temperatures. They have some clear advantages: PEM electrolysers are compact, produce very pure hydrogen, and perform quicker than AEMs, with higher flexibility. However, they’re facing a big barrier: some of their parts, commonly catalysts, require precious metals like platinum and iridium, which increases the cost and compromises sustainability in the long term.
How do electrolysers contribute to the decarbonisation of the European economy?
Now that you know all about the technology, you may wonder how advanced it really is. We’ve examined the latest reports from the International Energy Agency (IEA) to have a global view. From their latest report on electrolysers, published in 2022:

So, out of the four electrolyser technologies we listed, only PEM and alkaline electrolysers are actually available on the market, while SOEC and AEM are still on less advanced, demonstrator phases. However, the amount of electrolyser capacity built throughout the world is rising, especially in the last two years.
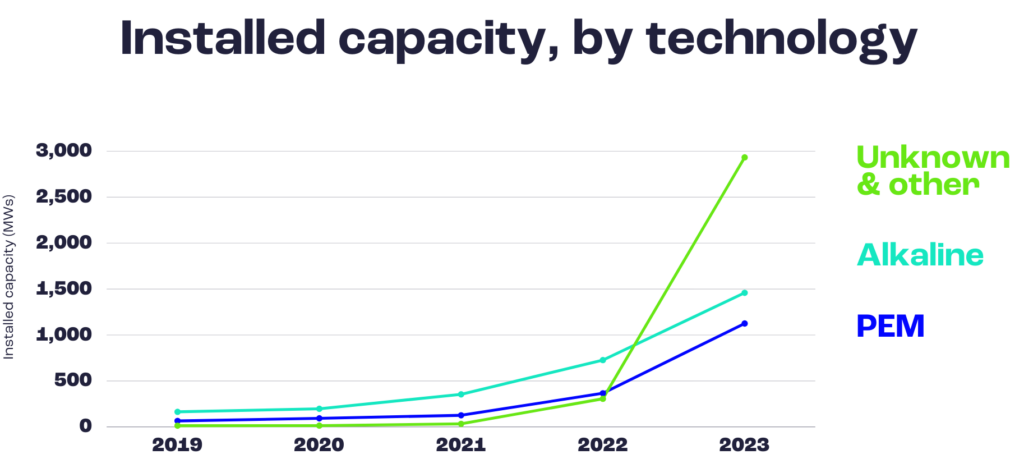
Therefore, it seems clear that electrolysers are taking off. Hydrogen production will play a special role in the decarbonisation of some specific sectors, especially those that usually showcase great difficulties in reducing emissions: like long-haul transportation and the manufacturing of key chemicals and raw materials such as iron and steel.
At ANEMEL, we’re improving electrolyser technology, to make green hydrogen from dirty waters, making green hydrogen cheaper and even more sustainable.
Additional reading and references
- https://ec.europa.eu/commission/presscorner/detail/es/ip_22_2829
- https://www.cummins.com/news/2020/11/16/electrolyzers-101-what-they-are-how-they-work-and-where-they-fit-green-economy
- https://www.energy.gov/eere/fuelcells/hydrogen-production-electrolysis
- https://www.carbonbrief.org/in-depth-qa-does-the-world-need-hydrogen-to-solve-climate-change/
- https://www.carbonbrief.org/guest-post-can-green-hydrogen-grow-fast-enough-for-1-5c/
- https://www.iea.org/fuels-and-technologies/hydrogen
- https://oficinac.es/informes-c/hidrogeno-verde
- https://www.iea.org/reports/electrolysers